COPD Management is Greater Than the Sum of Its Parts
Posted on January 23, 2022 |
This post was authored by Michael W. Hess, MPH, RRT, RPFT, Senior Director of Public Outreach & Education, COPD Foundation
“When all you have is a hammer, every problem looks like a nail.”
It can be amazingly easy to get tunnel vision as a health care specialist. One can get so used to treating certain conditions that the care of every person in your care gets viewed through the lens of that particular pathology. It is a lesson I myself have learned the hard way as a respiratory therapist, especially as one working in chronic disease management. I remember one specific patient who first presented with spirometry, symptoms, and imaging entirely consistent with emphysema. We started them on a particular medication regimen and soon got their symptoms under control, for which they were quite grateful. Then one day they came back, complaining of new-onset cough. The rest of his primary care team and I worked diligently over the course of the next few months, following right along with best practices and strategy recommendations, adding this medication or that, calling in airway consults, and various other tricks to address this ‘progression’ of COPD. Unfortunately, not a single thing worked for this person, and while their shortness of breath and activity tolerance were still great, the cough was more bothersome every day. We were stumped.
Fortunately, our person ended up having to visit a gastroenterologist for an unrelated problem. They were finally able to diagnose a hiatal hernia, which was causing frequent reflux, including during sleep. This was, in turn, continually irritating the airways, causing an increase in mucus production and therefore cough throughout the day. Within the next few months, surgery was scheduled, the hernia was repaired, and virtually overnight our patient was able to go from a maxed-out regimen with diminishing hope back to their original basic bronchodilator monotherapy.
While we were able to celebrate a happy ending, we also had to start questioning ourselves. How did we miss that? How did we make this poor person go through months of decreasing quality of life? Most importantly, how do we make sure that does not happen to others? While the answers to these questions are never simple, they are important to consider. After all, this patient was hardly unique; studies suggest 80-90% of people diagnosed with COPD have at least one other concurrent condition,1 and as many as half have two or more. That means your people with COPD aren’t just people with COPD, and the more conditions one has, the more likely they are to have heavier symptom burden.2 Treating those additional conditions is essential for optimal outcomes.
"Various studies have looked at the burden of concurrent conditions and their effects on COPD, both morbidity and mortality, culminating in the development of the COPD “comorbidome.3”
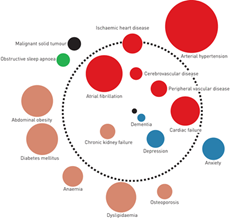
This diagram gives insight into a variety of conditions that affect and are affected by COPD, from the obvious (heart disease, lung cancer) to the less so (obesity and osteoporosis). However, it’s not necessarily complete, nor does it give insight into how these conditions can affect our COPD population. That’s where clinical expertise comes in, with a touch of mindfulness.
Perhaps one of the best examples of this is the overlap between bronchiectasis and COPD. Under many circumstances, the symptoms of chronic bronchitis can mask the onset of bronchiectasis to the point where approximately half of all those with at least moderate COPD symptoms are thought to have bronchiectasis.4 It can also be unclear which condition is the proverbial chicken and which is the egg, with one study indicating over 40% of people with COPD end up developing bronchiectasis within 7 years.5 That points to the importance of recognizing symptoms like intractable cough and recurrent pulmonary infections early on, and adding diagnostic support like CT imaging to better understand what’s actually happening inside the lung.
Detailed investigation can help identify a variety of these ‘sleeper’ conditions and more accurately guide clinical decision-making. One of the more insidious causes of bronchiectasis is infection with one of the family of pathogens known as nontuberculosis mycobacteria, or NTM. Most people are exposed to NTM on a near-constant basis, owing to their ubiquity in environments around the globe. However, in people with weakened immune and/or respiratory systems (like those with COPD), they can opportunistically cause serious complications. And, because those complications can be relatively nonspecific or align with some already in place, NTM lung disease can go undetected long enough to cause significant additional damage. That damage can be worsening bronchiectasis, it can be nodular development throughout the lung, or it can even appear as cavitary lesions, depending on age, biological sex, and specific pathogen.6 Recognizing NTM lung disease, by CT imaging and sputum or bronchial culture samples, is essential to developing a monitoring and treatment plan for people at risk for worsening progression.7
We commonly caution people about losing the ability to see the forest for the trees, meaning that it’s easy to get that tunnel vision that forces us to focus on individual aspects of a problem rather than the overall picture. But it can also be all too easy for that big picture to obscure all the factors that contribute to someone’s case. It is essential for clinicians to find a middle way there, in order to save their patients the trouble of trying to drive in a screw with a hammer.
References
- Kiani FZ, Ahmadi A. Prevalence of different comorbidities in chronic obstructive pulmonary disease among Shahrekord PERSIAN cohort study in southwest Iran. Sci Rep. 2021;11(1):1-8. https://www.nature.com/articles/s41598-020-79707-y
- Raherison C, Ouaalaya EH, Bernady A, et al. Comorbidities and COPD severity in a clinic-based cohort. BMC Pulm Med. 2018;18(1):1-10. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6048834/
- Almagro P, Cabrera FJ, Diez-Manglano J, et al. Comorbidome and short-term prognosis in hospitalised COPD patients: The ESMI study. Eur Respir J. 2015;46(3):850-853. https://erj.ersjournals.com/content/46/3/850
- Ni Y, Shi G, Yu Y, Hao J, Chen T, Song H. Clinical characteristics of patients with chronic obstructive pulmonary disease with comorbid bronchiectasis: A systemic review and meta-analysis. Int J COPD. 2015;10(1):1465-1475. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4524532/
- Martínez-Garciá MÁ, De La Rosa-Carrillo D, Soler-Cataluña JJ, et al. Bronchial Infection and Temporal Evolution of Bronchiectasis in Patients with Chronic Obstructive Pulmonary Disease. Clin Infect Dis. 2021;72(3):403-410. https://pubmed.ncbi.nlm.nih.gov/31967312/
- Griffith DE. Overview of nontuberculous mycobacterial infections. UpToDate.com. Published online 2020:2-11. Accessed January 18, 2022. https://www.uptodate.com/contents/overview-of-nontuberculous-mycobacterial-infections?search=ntm lung
- Daley CL, Iaccarino JM, Lange C, et al. Treatment of nontuberculous mycobacterial pulmonary disease: An official ATS/ERS/ESCMID/IDSA clinical practice guideline. Clin Infect Dis. 2020;71(4):E1-E36. https://academic.oup.com/cid/article/71/4/e1/5867961?login=true
- Zhang P, Chen B, Lou H, et al. Predictors and outcomes of obstructive sleep apnea in patients with chronic obstructive pulmonary disease in China. BMC Pulm Med. 2022;22(1):1-11. https://bmcpulmmed.biomedcentral.com/articles/10.1186/s12890-021-01780-4
- McNicholas WT. COPD-OSA Overlap Syndrome: Evolving Evidence Regarding Epidemiology, Clinical Consequences, and Management. Chest. 2017;152(6):1318-1326. https://journal.chestnet.org/article/S0012-3692(17)30742-0/fulltext